written by
Subscribe to our monthly water newsletter
We might send you an email if we have something news worthy, written by our water professionals
Dissolved Oxygen and Henry’s Law
To understand why warm water holds less oxygen, we begin with Henry’s Law, a principle of physical chemistry. It states:
The amount of gas dissolved in a liquid is proportional to the gas’s partial pressure and inversely affected by temperature.
In simpler terms:
The cooler the water, the easier it is for oxygen molecules in the air to dissolve.
The warmer the water, the harder it is for oxygen to stay in solution.
This means that temperature directly controls gas solubility, and oxygen is no exception.
Increased Molecular Activity in Warm Water
At higher temperatures:
Water molecules move faster.
They collide more frequently.
They form fewer stable molecular “pockets” capable of holding dissolved gases.
Oxygen dissolved in water is held by weak physical forces. When molecular motion increases:
These forces are disrupted.
Oxygen molecules are pushed out of solution.
They escape back into the air.
This is why, for example:
Cold mountain streams are rich in oxygen.
Warm stagnant ponds often suffer from oxygen deficiency.
Warm Water Accelerates Gas Escape
When water is heated, gas solubility decreases not only because oxygen dissolves less readily but also because:
Dissolved oxygen diffuses out of the water more quickly.
Higher temperatures mean higher evaporation and surface agitation.
Oxygen molecules have a greater tendency to leave the liquid environment.
This double effect — poorer solubility and faster escape — explains why DO drops sharply in warm conditions.
Higher Temperatures Increase Biological Oxygen Demand
Not only does warm water contain less dissolved oxygen — life inside it uses oxygen faster, creating a double stress.
Warmer temperatures accelerate the metabolism of:
Fish
Microorganisms
Plants
Algae
As metabolic rates increase:
Respiration consumes more oxygen.
Decomposition speeds up.
Microbes multiply more rapidly.
So, in warm water:
Less oxygen is available.
More oxygen is being consumed.
This imbalance can lead to rapid DO depletion, especially in closed or nutrient-rich systems.
Effects in Agriculture and Hydroponics
In irrigation and hydroponic reservoirs, high water temperature can create a cascade of negative effects:
Lower DO Weakens Roots
Plant roots rely on oxygen for respiration. When DO drops:
Nutrient uptake decreases.
Protective cell functions weaken.
Low Oxygen Encourages Harmful Microbes
Warm, oxygen-poor water is the ideal environment for pathogens like:
Certain anaerobic bacteria
The combination of warm water and low DO significantly increases the risk of root rot.
Water Quality Declines
Warmer water encourages:
Algae blooms
Biofilm buildup
Higher microbial loads
All of these consume oxygen, worsening the problem.
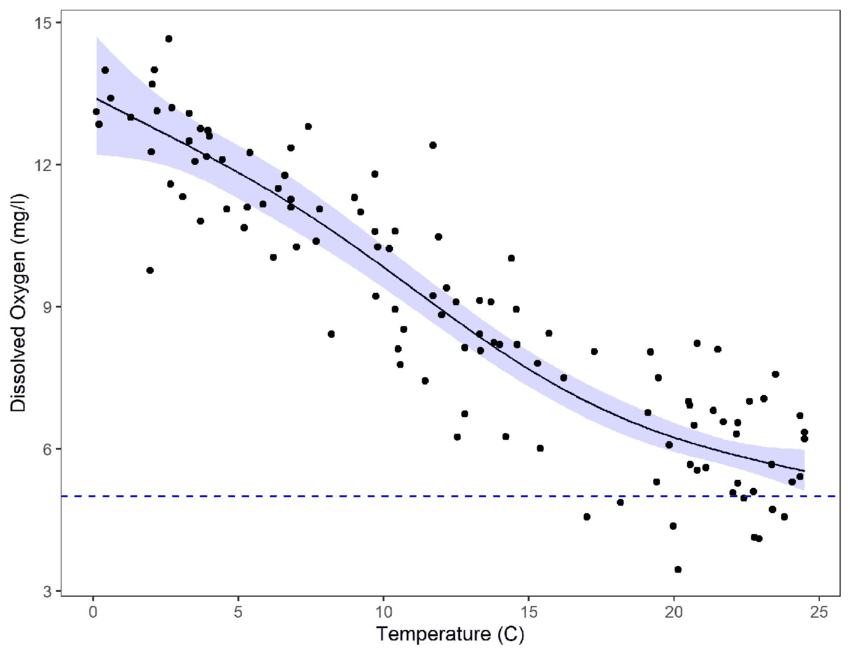
How Much Difference Does Temperature Make?
Although exact values depend on salinity and pressure, typical freshwater examples are:
Water Temperature | Approx. DO Saturation |
|---|---|
10°C (50°F) | ~11 mg/L |
20°C (68°F) | ~9 mg/L |
30°C (86°F) | ~7 mg/L |
By simply increasing water temperature from 20°C to 30°C, oxygen capacity can drop by 25% or more, before biological demand is even considered.
Cooling Water Improves DO Naturally
Unlike adding nutrients or chemicals, improving DO through cooling is simple and effective:
No artificial additives
No pumps required (though aeration helps)
More stable root and microbial environments
Even a small temperature reduction of 2–5°C (4–9°F) can significantly increase dissolved oxygen and reduce plant stress.
Conclusion
Warm water holds less dissolved oxygen because:
Higher temperatures reduce gas solubility according to Henry’s Law.
Increased molecular movement physically pushes oxygen out of solution.
Warmer conditions accelerate oxygen consumption by living organisms.
This combination makes warm water a high-risk environment for oxygen depletion, plant stress, and pathogen development. Keeping irrigation or hydroponic water cool — often below 22–24°C (72–75°F) — helps maintain healthy dissolved oxygen levels, supports stronger roots, and reduces disease risk naturally.
At Waboost we specialize at adding dissolved oxygen to the water as efficiently as possible. Check out our page about nanobubbles to learn more.




